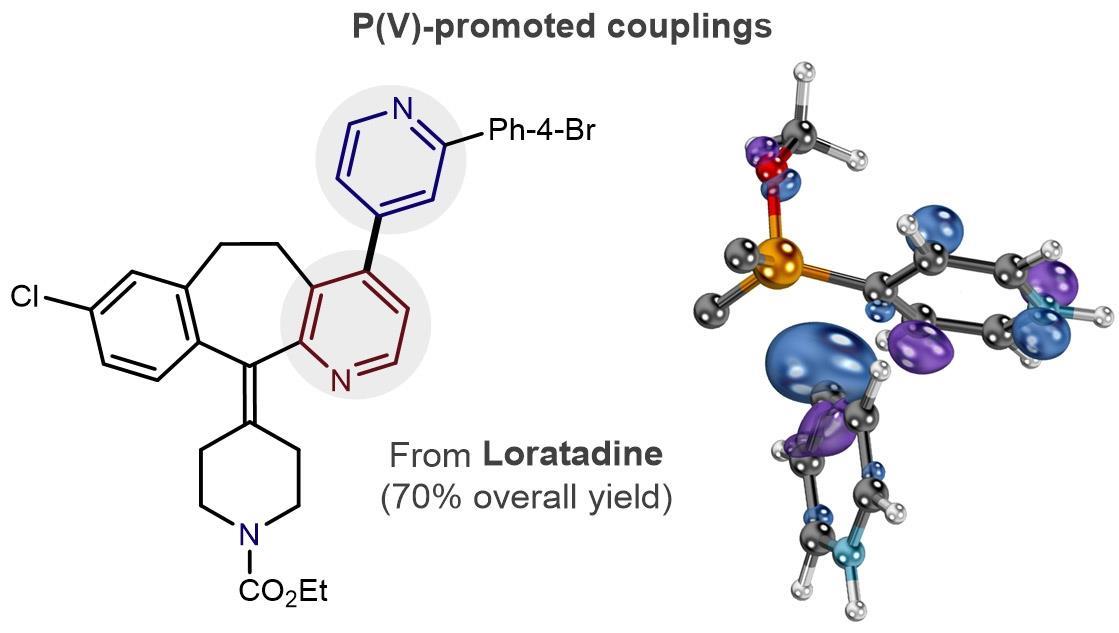
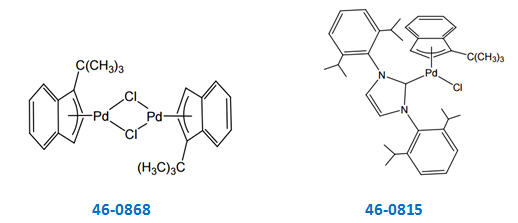
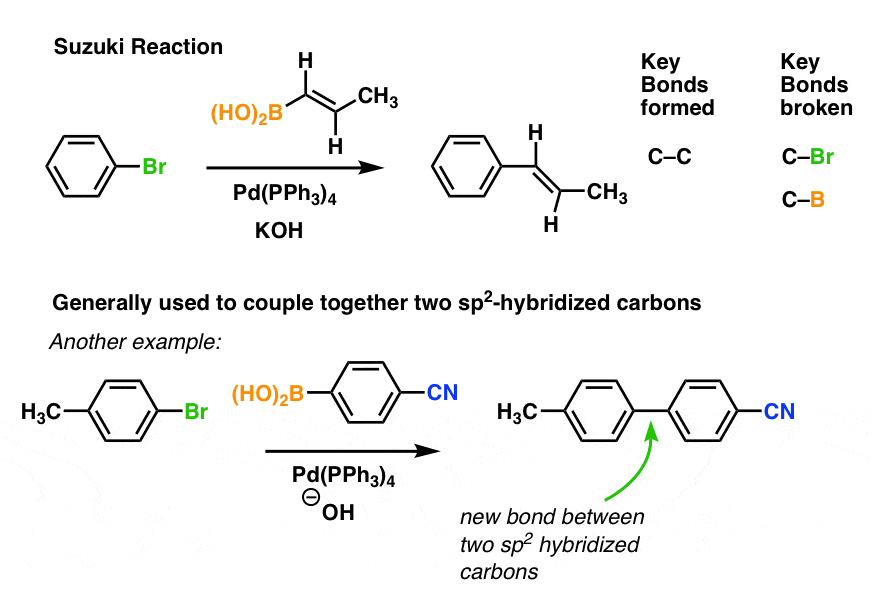
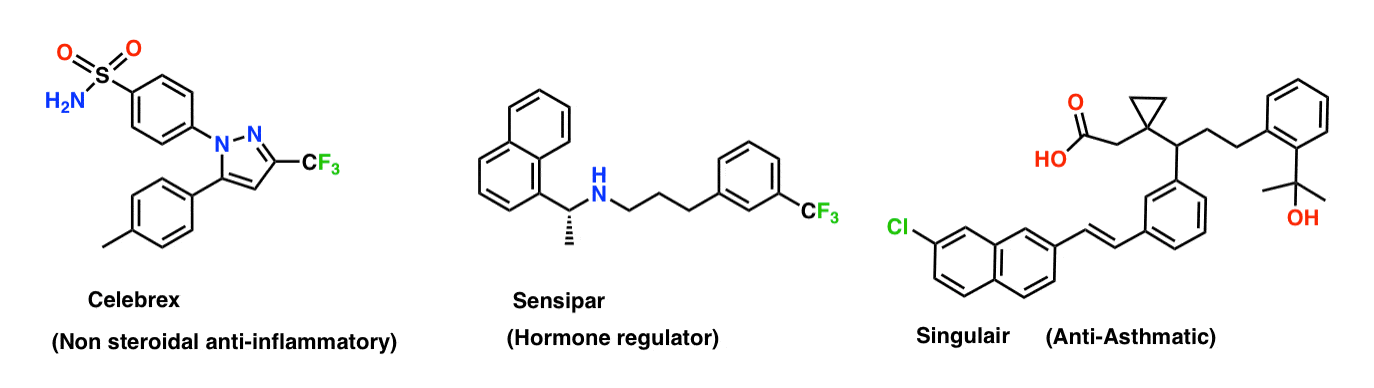
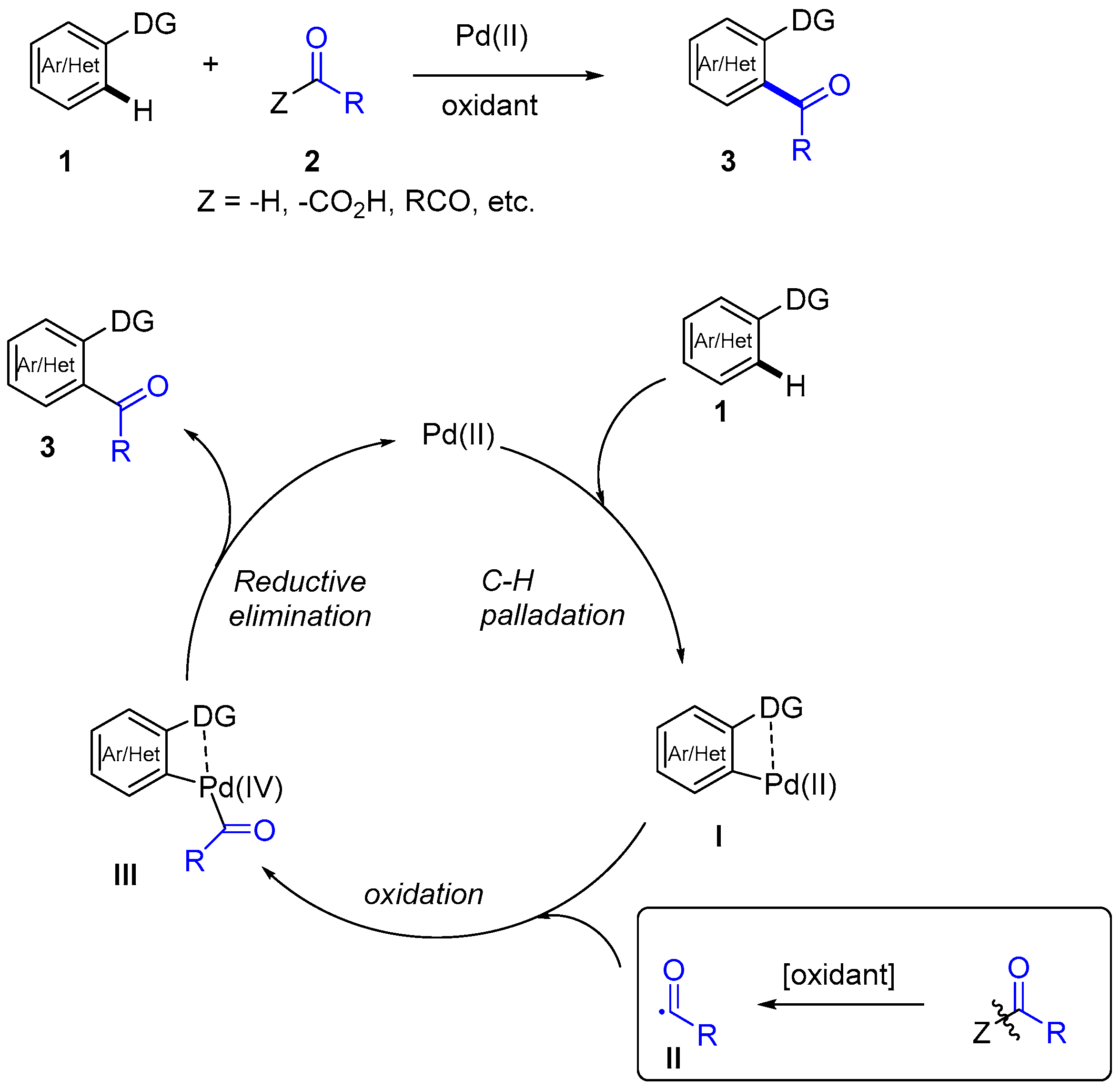
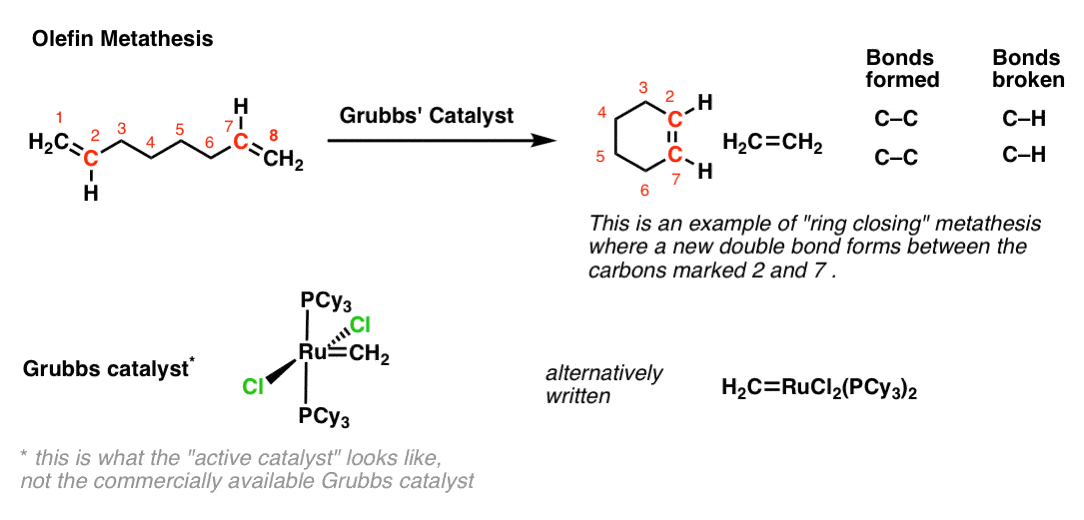
Palladium-Catalyzed, Ring-Forming Aromatic C–H Alkylations with Unactivated Alkyl Halides | Journal of the American Chemical Society

Application of Palladium-Catalyzed C(sp2)–H Bond Arylation to the Synthesis of Polycyclic (Hetero)Aromatics - ScienceDirect

Cyclic Ether Synthesis via Palladium-Catalyzed Directed Dehydrogenative Annulation at Unactivated Terminal Positions | Journal of the American Chemical Society

Quadruple C-H activation coupled to hydrofunctionalization and C-H silylation/borylation enabled by weakly coordinated palladium catalyst | Nature Communications

Palladium‐Catalyzed Regioselective and Stereospecific Ring‐Opening Suzuki‐Miyaura Arylative Cross‐Coupling of 2‐Arylazetidines with Arylboronic Acids - Takeda - 2021 - Advanced Synthesis & Catalysis - Wiley Online Library

Catalytic asymmetric C–Si bond activation via torsional strain-promoted Rh-catalyzed aryl-Narasaka acylation | Nature Communications

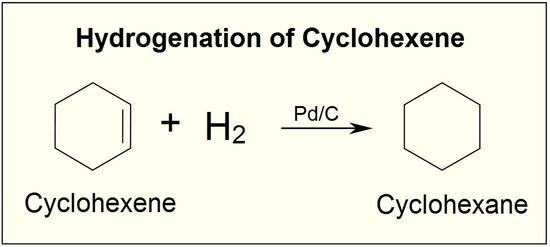
Compound A has the formula C_8H_8. It reacts rapidly with KMnO_4 to give CO_2 and a carboxylic acid, B (C_7H_6O_2) but reacts with only 1 molar equivalent of H_2 on catalytic hydrogenation

Palladium-Catalyzed Ring-Closing Reaction via C–N Bond Metathesis for Rapid Construction of Saturated N-Heterocycles | Journal of the American Chemical Society

Recoverable polystyrene-supported palladium catalyst for construction of all-carbon quaternary stereocenters via asymmetric 1,4-addition of arylboronic acids to cyclic enones - ScienceDirect

The Emergence of Palladium‐Catalyzed C(sp3)−H Functionalization of Free Carboxylic Acids - Das - 2021 - Chemistry – An Asian Journal - Wiley Online Library
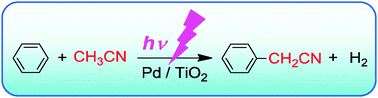
A heterogeneous palladium catalyst hybridised with a titanium dioxide photocatalyst for direct C–C bond formation between an aromatic ring and acetonitrile - Chemical Communications (RSC Publishing)

Catalytic cycle of the Pd-promoted carbon-carbon (C-C) bondformation... | Download Scientific Diagram

The Development of Efficient Protocols for the Palladium-Catalyzed Cyclization Reactions of Secondary Amides and Carbamates

Application of Palladium-Catalyzed C(sp2)–H Bond Arylation to the Synthesis of Polycyclic (Hetero)Aromatics - ScienceDirect

The application of a supported palladium catalyst for the hydrogenation of aromatic nitriles - ScienceDirect
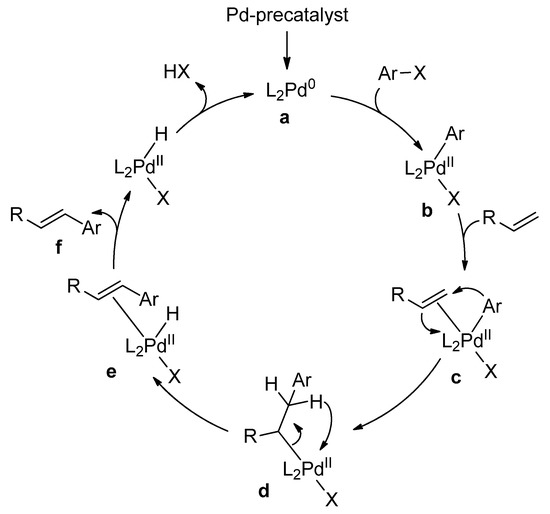
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond